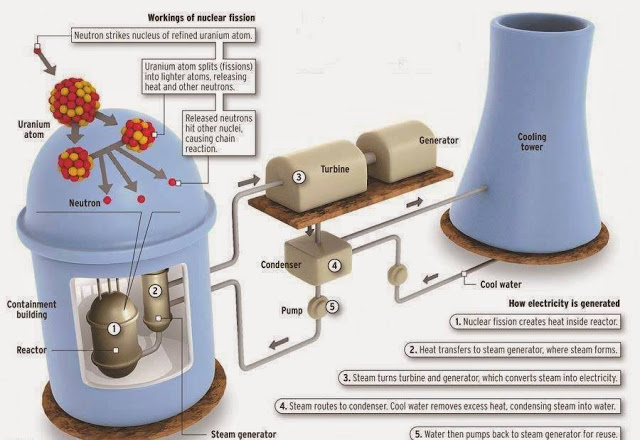
The precise daughter products created in fission can't be accurately predicted, as the process is subject to a high degree of chance and variation. The rest of the energy is transferred as kinetic energy to the released neutrons or carried away by high-energy radiation in the form of gamma rays. This is when the term c² becomes important as this tells us that even a tiny amount of mass liberates a lot of energy.Īround 85 percent of this energy liberated in fission reactions is released as kinetic energy granted to the daughter nuclei. The difference in mass between the starting particle and its daughter particles is tiny - about 0.1 percent of the original mass. When a fissile material absorbs a neutron and breaks apart, the mass going into the reaction is slightly higher than the mass than emerges from it. In its simplest form, this is encapsulated by arguably the world's most famous equation: energy equals mass times the speed of light squared or e=mc². The reason this process releases energy is related to Albert Einstein's discovery that mass and energy are interchangeable. (Image credit: Peter Hermes Furian/Getty Images) That means by the tenth 'generation,' there are 1,024 fissions, and by generation 80 there are 6 x 10²³ fission reactions.Īn illustration of the chain reaction underlying nuclear fission. This leads to a chain reaction of splitting nuclei, producing a doubling of fission reactions each time a nucleus is split. Those neutrons are then ejected, striking other nuclei. The released neutrons traveling at a speed of around 33 million feet per second (10 million meters per second, or about three percent of the speed of light) - go on to strike two more nuclei, causing them to split and release four neutrons. The daughter particles are rapidly pushed apart as a result of their positive charges repelling one another. The gap between the lobes created by neutron capture eventually exceeds the point at which the strong nuclear force - which binds protons and neutrons together in the atomic nucleus and is powerful only across tremendously small ranges - can hold them together.Īs a result, the nucleus fractures into smaller fragments , usually around half the mass of the starting particle, also releasing at least two, sometimes three, neutrons. The energy needed to kick start fission is around 7 to 8 million electronvolts (MeV), and when a neutron carrying this level of energy or more strikes the target nucleus, the energy it imparts deforms the nucleus into a double-lobed peanut-like shape. In nuclear reactors, this is an isotope - an atom with a different neutron count in its nucleus - of the heavy elements uranium or plutonium. Induced nuclear fission occurs when a particle - commonly a neutron - passes a large target atomic nucleus and is captured by it. 
(Image credit: Dorling Kindersley/Getty Images) How does nuclear fission produce energy? In 1933, Hungarian physicist Leó Szilárd first formalized the idea that neutron-driven fission of heavy atoms could be used to create a nuclear chain reaction having generated energy by using protons to split lithium the year before.įinally, in December 1938, physicists Lise Meitner and Otto Frisch realized that isotopes of barium that appeared mysteriously during neutron-uranium bombarding experiments conducted by colleague Otto Hann were the result of the uranium nuclei undergoing fission.Īn illustration of how fission works, depicting uranium 235 nucleus splitting into fragments and creating new nuclei. Two years after the discovery of the neutron in 1932 by James Chadwick, Enrico Fermi and his colleagues in Rome began pelting these newly found particles at uranium with other physicists also reaching the conclusion the particle would make a good probe of the atomic nucleus. This led to the discovery by Henri Becquerel, Marie Curie, Pierre Curie, and Rutherford that the atoms of elements could 'decay' and transmute to another element via the emission of an alpha particle.  
The discovery of induced fission wouldn't have been possible without the strides made by Ernest Rutherford and Niels Bohr toward a coherent picture of the atom during the 1910s. The detonation of so-called 'atom bombs' is signified by the sight of a mushroom cloud - a dreadful reminder of the power of the atom and of fission itself. When this process is allowed to run unchecked, however, it gives rise to a powerful and destructive force.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
